Free Quality Assurance Audit Checklist
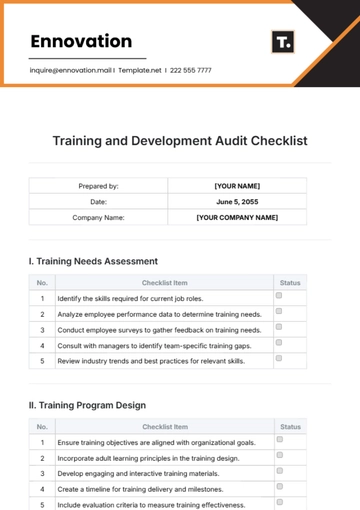
Prepared by: | [YOUR NAME] |
Date: | May 19, 2055 |
Company Name: | [YOUR COMPANY NAME] |
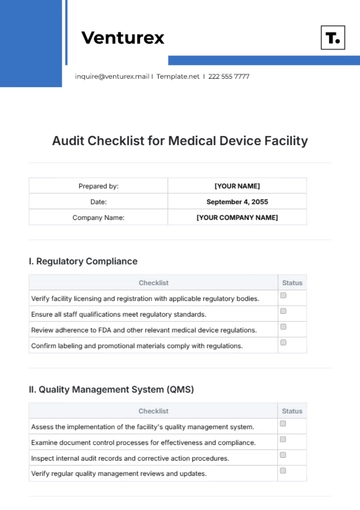
I. Documentation Review
Status | Task |
|---|---|
Review the QA policy and procedures for adequacy and compliance with standards. | |
Ensure all process manuals and SOPs (Standard Operating Procedures) are current and complete. | |
Verify records of training for relevant personnel are maintained accurately. | |
Check documentation for non-conformance management and corrective actions. |
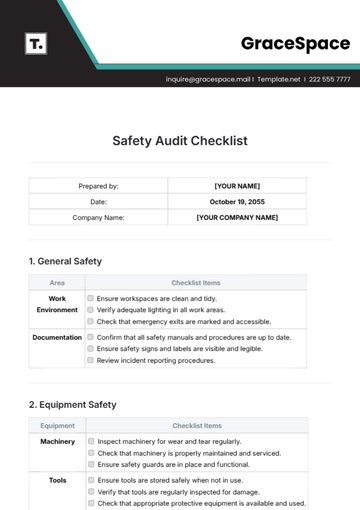
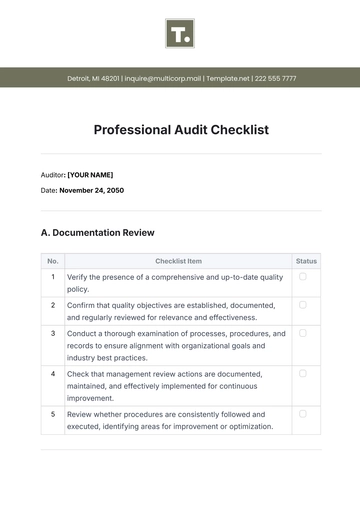
II. Process Control
Status | Task |
|---|---|
Monitor compliance with approved processes and quality plans. | |
Assess process monitoring and measurement techniques for effectiveness. | |
Evaluate change control processes and their implementation across operations. | |
Check for consistency in quality checks and in-line inspections. |
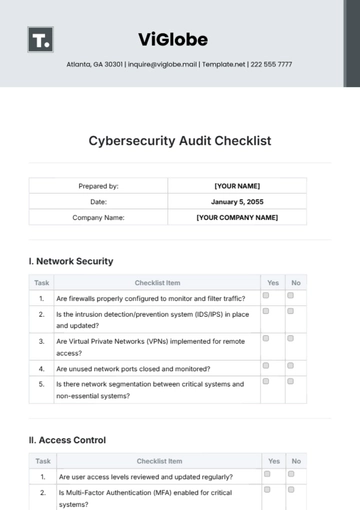
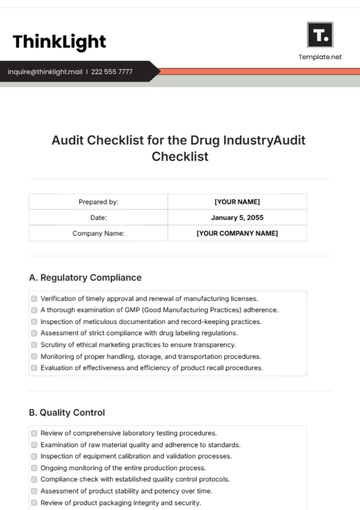
III. Product Verification
Status | Task |
|---|---|
Validate that products meet specified quality requirements before shipment. | |
Examine testing methods and equipment calibration for accuracy. | |
Inspect storage conditions for compliance with specified parameters. | |
Review quality control logs and rejection rates for trends and issues. |
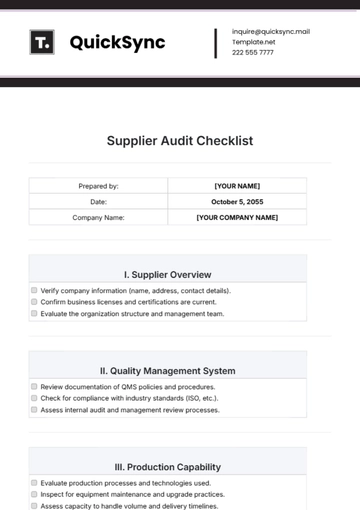
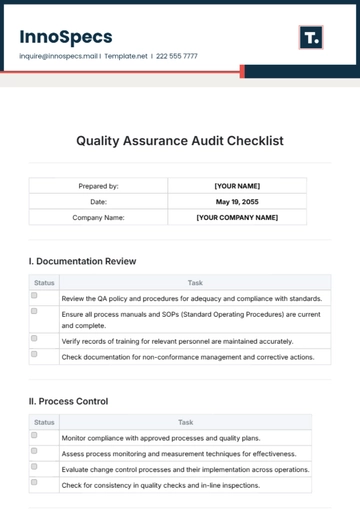
IV. Continuous Improvement
Status | Task |
|---|---|
Analyze data from quality metrics to identify improvement opportunities. | |
Review the effectiveness of corrective and preventive actions taken. | |
Check onboarding and regular training programs for staff development. | |
Ensure open communication channels for feedback and suggestions. |
- 100% Customizable, free editor
- Access 1 Million+ Templates, photo’s & graphics
- Download or share as a template
- Click and replace photos, graphics, text, backgrounds
- Resize, crop, AI write & more
- Access advanced editor
Ensure top-notch product and service quality with Template.net's Quality Assurance Audit Checklist Template. This fully customizable and editable template provides a systematic approach to quality audits, allowing businesses to uphold their commitment to excellence. Tailor the checklist effortlessly with our user-friendly AI Editor Tool to match your industry and specific quality standards.
You may also like
- Cleaning Checklist
- Daily Checklist
- Travel Checklist
- Self Care Checklist
- Risk Assessment Checklist
- Onboarding Checklist
- Quality Checklist
- Compliance Checklist
- Audit Checklist
- Registry Checklist
- HR Checklist
- Restaurant Checklist
- Checklist Layout
- Creative Checklist
- Sales Checklist
- Construction Checklist
- Task Checklist
- Professional Checklist
- Hotel Checklist
- Employee Checklist
- Moving Checklist
- Marketing Checklist
- Accounting Checklist
- Camping Checklist
- Packing Checklist
- Real Estate Checklist
- Cleaning Checklist Service
- New Employee Checklist
- Food Checklist
- Home Inspection Checklist
- Advertising Checklist
- Event Checklist
- SEO Checklist
- Assessment Checklist
- Inspection Checklist
- Baby Registry Checklist
- Induction Checklist
- Employee Training Checklist
- Medical Checklist
- Safety Checklist
- Site Checklist
- Job Checklist
- Service Checklist
- Nanny Checklist
- Building Checklist
- Work Checklist
- Office Checklist
- Training Checklist
- Website Checklist
- IT and Software Checklist
- Performance Checklist
- Project Checklist
- Startup Checklist
- Education Checklist
- Home Checklist
- School Checklist
- Maintenance Checklist
- Planning Checklist
- Manager Checklist
- Wedding Checklist
- Vehicle Checklist
- Travel Agency Checklist
- Vehicle Inspection Checklist
- Interior Design Checklist
- Backpacking Checklist
- Business Checklist
- Legal Checklist
- Nursing Home Checklist
- Weekly Checklist
- Recruitment Checklist
- Salon Checklist
- Baby Checklist
- Equipment Checklist
- Trade Show Checklist
- Party Checklist
- Hospital Bag Checklist
- Evaluation Checklist
- Agency Checklist
- First Apartment Checklist
- Hiring Checklist
- Opening Checklist
- Small Business Checklist
- Rental Checklist
- College Dorm Checklist
- New Puppy Checklist
- University Checklist
- Building Maintenance Checklist
- Work From Home Checklist
- Student Checklist
- Application Checklist